Share
Report
Question
Explanation :
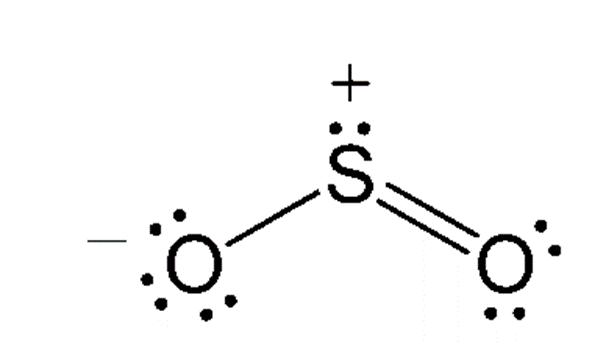
- It is a bent shape molecule with bond order 1.5.
- It has a bond angle (119)∘.
- Due to lone pair lone pair repulsion bond angle reduced to (119)∘from 120∘.
- Sulphur is sp2 hybridized in it.
Final Answer: Hence Sulphur has bent shape structure
solved
5
wordpress
4 mins ago
5 Answer
70 views
+22

Leave a reply